In the ground state, they are arranged in the electron configuration 3s 23p 2. For example, a silicon atom has fourteen electrons. In electrical insulators and semiconductors, the valence band is the highest range of electron energies in which electrons are normally present at absolute zero temperature. In solid-state physics, the valence band and conduction band are the bands closest to the Fermi level and thus determine the electrical conductivity of the solid. To understand the difference between metals, semiconductors and electrical insulators, we have to define the following terms from solid-state physics: from ionizing radiation) to cross the band gap and to reach the conduction band. In contrast to conductors, electrons in a semiconductor must obtain energy (e.g. In solid-state physics, this energy gap or band gap is an energy range between valence band and conduction band where electron states are forbidden. They have an energy gap less than 4eV (about 1eV). The name semiconductor comes from the fact that these materials have an electrical conductivity between that of a metal, like copper, gold, etc. Semiconductors are materials, inorganic or organic, which have the ability to control their conduction depending on chemical structure, temperature, illumination, and presence of dopants. Insulators, on the other hand, are made of a wide variety of materials depending on factors such as the desired resistance. Conductors are made of high-conductivity materials such as metals, in particular copper and aluminium. Substances in which electricity can flow are called conductors. While resistivity is a material property, resistance is the property of an object. Electrical resistance is expressed in Ohms. Note that, electrical resistivity is not the same as electrical resistance. The SI unit of electrical resistivity is the ohm-metre (Ω⋅m). The symbol of resistivity is usually the Greek letter ρ (rho). 
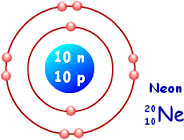
A low resistivity indicates a material that readily allows the flow of electric current. m.Įlectrical resistivity and its converse, electrical conductivity, is a fundamental property of a material that quantifies how strongly it resists or conducts the flow of electric current.Because shell two has eight electrons it is now full.Neon – Electrical Resistivity and Electrical ConductivityĮlectrical resistivity of Neon is - nΩ Looking at the picture, you can see there are two electrons in shell one and eight in shell two. That means there are 10 electrons in a neon atom. for the element of NEON, you already know that the atomic number tells you the number of electrons. Once one shell is full, the next electron that is added has to move to the next shell. As you learn about elements with more than eighteen electrons you will find that shell three can hold more than eight. Shell number one can only hold 2 electrons, shell two can hold 8, and for the first eighteen elements shell three can hold a maximum of eight electrons. The electrons like to be in separate shells/orbitals. In an atom, the electrons spin around the center, also called the nucleus. Each of those colored balls is an electron. In the next section we're going to cover electron orbitals or electron shells. It tells you the mass of one atom, how many pieces are inside, and where it should be placed on the periodic table. That box on the left has all of the information you need to know about one element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
